|
In contrast, copper has a specific heat capacity of 0.39 J. 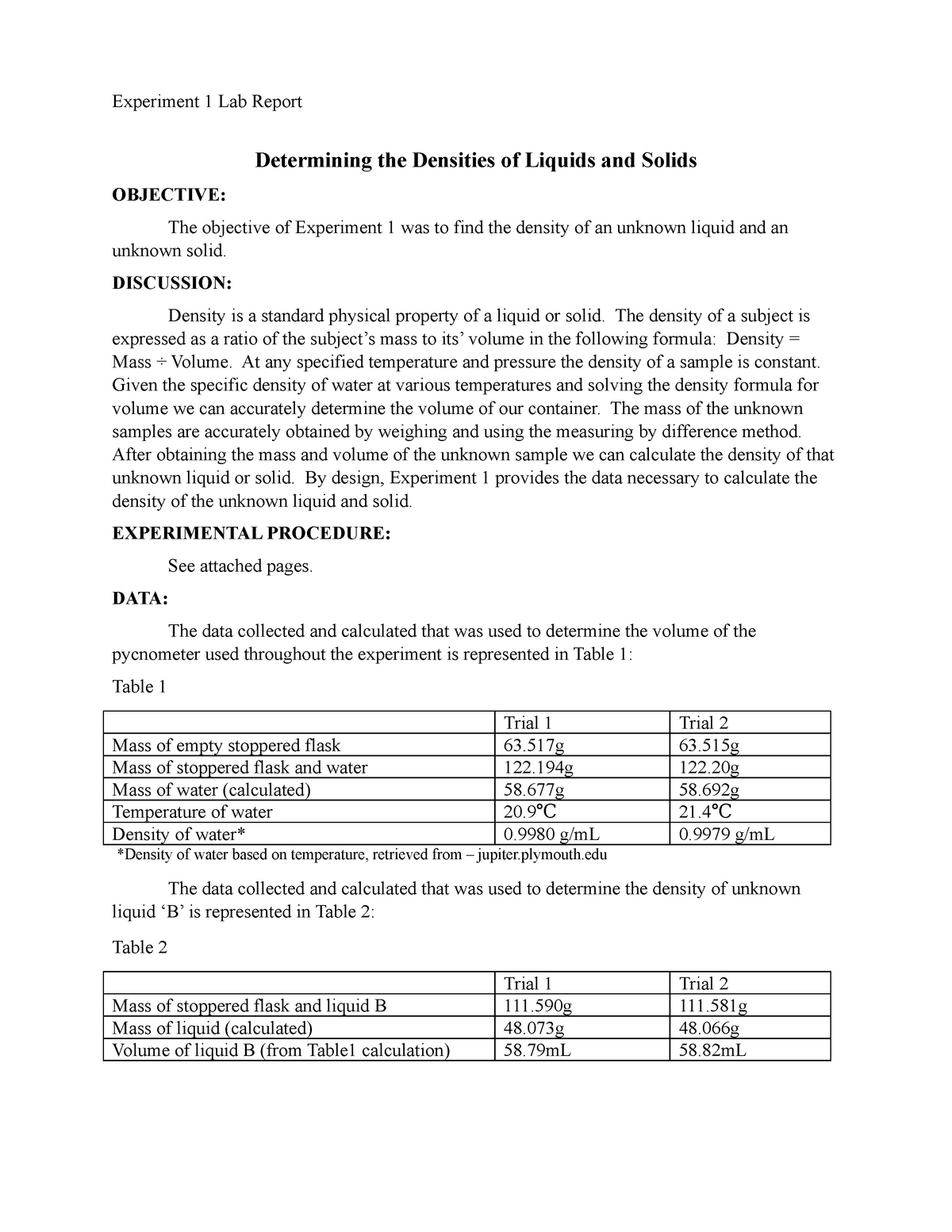
This is a much higher value than that of most other substances, which makes water exceptionally good at regulating temperature. Water has a specific heat capacity of 4.18 J (or 1 calorie/gram ☌). It is applicable to wetlands, beaches, boats, sewage and other places. It evaporates sewage, salt water and other watery objects by the use of heat generated from sunlight. What is C of water?Īccording to its creator, the C-Water ” is a device which produces freshwater. As a result, water plays a very important role in temperature regulation. The specific heat of water is 1 calorie/gram ☌ = 4.186 joule/gram ☌ which is higher than any other common substance. 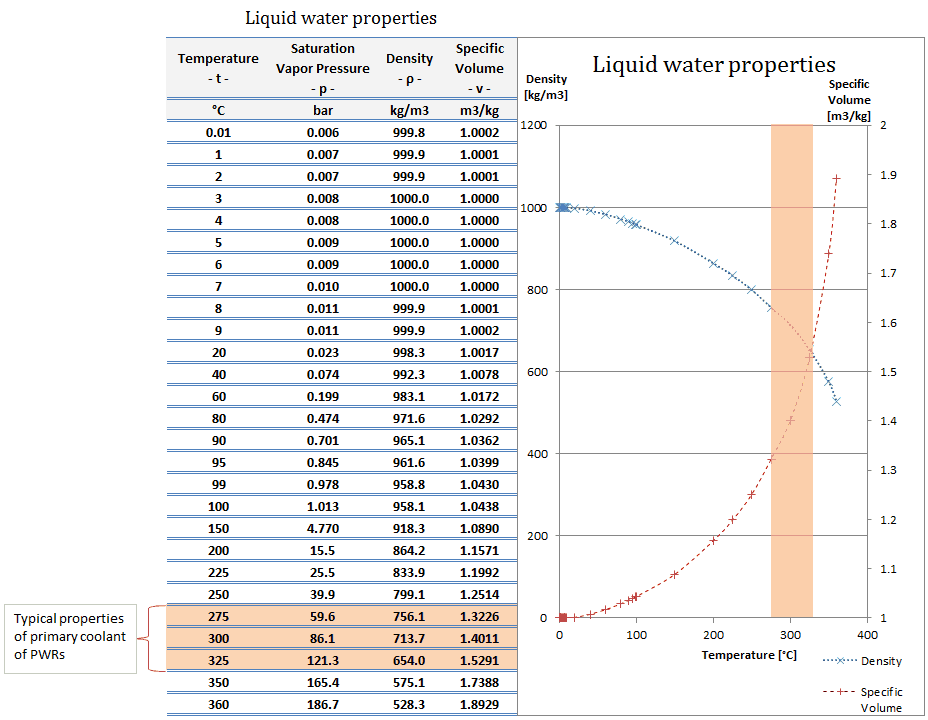
› Water and steam properties calculatorįrequently Asked Questions What is the specific heat of water?.› Property table calculator thermodynamics.› Thermophysical properties of water table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
